pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
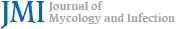
pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Sooyeong Kim,Ji Ho Park,Kwang Eon Han,Eun Jung Choi,Hyun-Chang Ko,Jung Hyo Ahn,Su-Jin Kim,Seung Min Lee,Sangwoo Moon,Tae-Sik Jang,Ji Eun Lee
10.17966/JMI.2026.31.1.18 Epub 2026 March 31
Abstract
Background: Fungal keratitis is a severe corneal infection that can lead to serious complications, including corneal perforation and endophthalmitis. Early identification of the causative fungi is essential for selecting appropriate antifungal therapy and improving clinical outcomes.
Objective: This study aimed to evaluate the efficacy of a culture method using a flocked swab combined with brain-heart infusion (BHI) liquid medium, compared with the conventional cotton swab method, in patients with fungal keratitis.
Methods: A retrospective review was performed on 15 patients clinically suspected of having fungal keratitis between March 2024 and July 2025. Each corneal lesion was sampled with a conventional cotton swab and a flocked swab before antifungal treatment. Conventional swabs were inoculated onto blood agar, chocolate agar, MacConkey agar, and Sabouraud dextrose agar plates, whereas flocked swabs were first incubated in BHI medium for 24 hours before being plated onto the same solid media.
Results: The culture positivity rate was 20.0% (3/15) with the conventional method and 86.7% (13/15) with the flocked swab method (p = 0.00195). The median time to positivity was significantly shorter with the flocked swab method (96 hours; IQR 72-96) than with the conventional method (116 hours; IQR 107-117; p = 0.016). Three patients (20.0%) had mixed bacterial-fungal infections, two of which were detected only by the flocked swab method.
Conclusion: The flocked swab with BHI liquid medium improved culture yield and reduced the time to positivity compared with the conventional cotton swab method, suggesting improved diagnostic performance.
Keywords
Brain-heart Infusion Flocked swab Fungal keratitis
Infectious keratitis is a significant cause of corneal blindness worldwide1. Without prompt and appropriate treatment, the disease may progress to severe complications, such as corneal perforation and endophthalmitis, and in advanced cases, it may even require enucleation2,3. The main risk factors for infectious keratitis include trauma, contact lens wear, preexisting ocular disease, and prior ocular surgery, while the causative organisms include bacteria, fungi, and, less commonly, Acanthamoeba4. Notably, fungal keratitis is associated with a higher rate of serious complications, and its disease course tends to be longer than that of other infectious etiologies5,6.
Although the symptoms and signs of fungal keratitis can provide valuable diagnostic clues, a definitive diagnosis requires microbiological confirmation through tests, such as potassium hydroxide (KOH) wet mount and culture7. Among these, fungal culture is the most sensitive method, playing a critical role not only in identifying the causative organisms but also in selecting appropriate antifungal therapy, ultimately improving treatment outcomes8,9. Conventional culture methods typic- ally involve obtaining corneal scrapings using a cotton swab or a blade8. However, the blade approach carries a risk of corneal perforation; therefore, the sampling technique must be chosen with caution10. The specimens collected from the corneal surface are inoculated onto selective media, such as Sabouraud dextrose agar plates, and culture results are usually confirmed 5-15 days later11.
Nevertheless, reported culture positivity rates for fungal keratitis vary widely across studies, ranging from approxi- mately 40% to over 80-15. This variability is influenced by factors, such as insufficient specimen volume, difficulties in the storage and transport of culture media, and the oper- ator's level of expertise16. Furthermore, false-negative results are common, even when fungal infection is clinically sus- pected17,18. Fungal keratitis is typically a low-biomass infection, and the limited number of viable organisms obtained from corneal lesions often results in low culture yield when using conventional sampling techniques10. Although molecular diagnostic methods are increasingly being adopted, fungal culture remains clinically relevant for guiding antifungal sus- ceptibility and detecting mixed bacterial–fungal infections. Therefore, optimizing culture-based diagnostic workflows continues to be important in clinical practice.
The flocked swab has been reported in both animal and human studies to provide superior specimen yield compared with conventional swabs. Previous studies have indicated that its uniformly arranged nylon fibers enhance both sample collection and release19,20. Building on these findings, our study applied this approach to human participants with fungal keratitis. Specifically, we compared the flocked swab culture method with the conventional method in terms of culture positivity and time to positivity, aiming to assess the clinical utility of the flocked swab culture for diagnosing fungal keratitis.
This study was approved by the Institutional Review Board of Pusan National University Yangsan Hospital (approval number: 55-2025-144) and was conducted in accordance with the principles of the Declaration of Helsinki.
We retrospectively reviewed the medical records of patients clinically suspected of having fungal keratitis at Pusan National University Yangsan Hospital between March 1, 2024, and July 31, 2025. Patients were included if fungi were identified on culture or if, despite negative culture results, they showed clinical improvement with antifungal therapy and were there- fore strongly suspected of having fungal keratitis. All corneal specimens were obtained before initiating empirical antibiotics or antifungal therapy. Patients initially suspected of having fungal keratitis but later diagnosed with nonfungal keratitis were excluded. This study aimed to compare the culture positivity rate and time to positivity between conventional and flocked swab methods in patients with fungal keratitis.
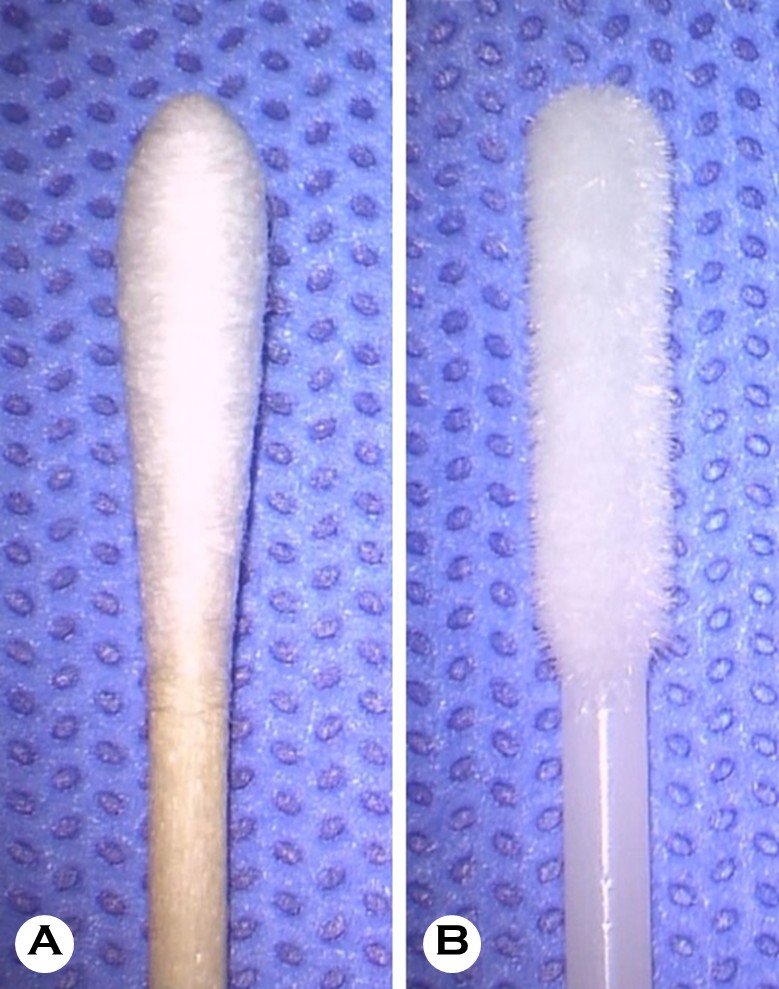
After topical anesthesia with 0.5% proparacaine hydro- chloride (Paracaine®, Hanmi Pharmaceutical, Seoul, Korea), corneal specimens were collected using both a conventional cotton swab (Fig. 1A) and a flocked swab (Fig. 1B). The con- ventional cotton swab was used first, followed by the flocked swab, with each swab sampling adjacent areas of the same lesion to minimize additional mechanical trauma. The cotton swab specimens were sealed in sterile tubes and immediately sent to the microbiology laboratory. They were streaked onto blood agar, chocolate agar, MacConkey agar, and Sabouraud dextrose agar plates (Fig. 2A). Sabouraud dextrose agar supplemented with chloramphenicol was used to suppress bacterial growth. Cycloheximide-containing media were not routinely employed, as cycloheximide can inhibit the growth of certain filamentous fungi relevant to fungal keratitis. Blood and chocolate agar plates were incubated at 35℃ under 5% CO2 conditions, while MacConkey agar plates were incubated at 35℃. Sabouraud dextrose agar plates were incubated aerobically at 25-30℃. Specimens collected with flocked swabs were immediately inoculated into brain-heart infusion (BHI) broth (Fig. 2B) and incubated at 27℃ for 24 hours, then streaked onto the same agar plates described above and incubated under the same conditions.
A positive culture was defined as the presence of at least one fungal colony on the inoculated Sabouraud agar plate. Cultures showing no fungal growth after up to 15 days of incubation were considered negative. Culture results were interpreted alongside clinical findings consistent with fungal keratitis. Fungal growth that corresponded with the typical clinical course and showed a good response to antifungal therapy was considered clinically significant. Fungal growth without clinical correlation was excluded and regarded as possible contamination. Time to positivity was defined as the interval between placing the inoculated plate in the incubator and the appearance of the first visible colony.
Statistical analyses were performed using IBM SPSS Statistics version 29 (IBM Corp., Armonk, NY, USA). Agreement be- tween the two culture methods was assessed using Cohen's kappa coefficient. In cases of disagreement, McNemar's test was applied to evaluate statistical significance. A p-value < 0.05 was considered statistically significant.
A total of 15 patients clinically suspected of having fungal keratitis were included in this study, comprising 10 men and 5 women. The mean age of the patients was 49.9 ± 23.2 years (range, 15-78 years), and the mean duration of illness before sample collection was 60.3 ± 121.1 days (range, 1-366 days). Infection was attributed to contact lens wear in 6 cases, ocular trauma in 3 cases, and unknown causes in 6 cases (Table 1). Specimen collection with either the con- ventional cotton swab or the flocked swab was well tolerated, with no procedure-related discomfort or adverse events.
|
Baseline characteristics |
Values |
|
Number, n |
15 |
|
Sex (Male/Female) |
10/5 |
|
Age (years) |
49.9±23.2 (15-78) |
|
Duration of illness (days) |
60.3±121.1 (1-366) |
|
Cause of infection |
|
|
Contact lens |
6 |
|
Trauma |
3 |
|
Unknown |
6 |
|
Values are presented as
mean ± standard deviation (range) or number, unless
otherwise indicated |
|
In culture results, the conventional cotton swab method yielded fungal growth in 3 of 15 cases (20.0%), whereas the flocked swab method showed positive results in 13 of 15 cases (86.7%), representing a significantly higher positivity rate (McNemar test, p = 0.00195) (Fig. 3). The two methods showed concordant results in 5 of 15 samples (33.3%), with 3 samples (20.0%) positive by both methods and 2 samples (13.3%) negative by both methods. Cohen's kappa coeffi- cient was κ = 0.01, indicating 'poor agreement' between the two methods. Discordant results were observed in 10 cases (66.7%), all of which were negative by the conventional method but positive by the flocked swab method. No samples were positive only with the conventional method and negative with the flocked swab method. This difference was statistically significant (McNemar test, p = 0.00195) (Table 2).
|
|
|
Flocked swab |
Total |
|
|
Positive culture |
Negative culture |
|||
|
Conventional |
Positive
culture |
n = 3 (20.0%) |
n = 0 (0.0%) |
n = 3 (20.0%) |
|
Negative
culture |
n = 10 (66.7%) |
n = 2 (13.3%) |
n = 12 (80.0%) |
|
|
|
Total |
n = 13 (86.7%) |
n = 2 (13.3%) |
n = 15 (100.0%) |
|
Cohen's kappa coefficient (κ) was calculated
to assess agreement between the two methods, and McNemar's test
was used to evaluate discordant results. Cohen's κ = 0.01, McNemar
test, p = 0.00195 |
||||
The median time to culture positivity was 116 hours (IQR, 107-117) for the conventional method and 96 hours (IQR, 72-96) for the flocked swab method. The flocked swab method yielded positive culture results significantly faster than the conventional method (Mann-Whitney U test, p = 0.016) (Fig. 4).
Among the 15 patients, mixed bacterial and fungal in- fections were identified in 3 (20.0%) cases (Table 3). Two cases (patients #3 and #13) were culture-negative by the conventional method but culture-positive by the flocked swab method, while one case (patient #10) was positive by both methods.
|
Patient
no. |
Pathogen |
Detection method |
|
|
Conventional |
Flocked swab |
||
|
#3 |
Bacterial + Fungal |
- |
+ |
|
#10 |
Bacterial + Fungal |
+ |
+ |
|
#13 |
Bacterial + Fungal |
- |
+ |
|
Others |
Fungal only |
|
|
In fungal keratitis, early identification of fungal pathogens is essential for selecting appropriate antifungal agents and initiating timely treatment. However, conventional culture methods are limited in clinical practice due to low sensitivity and prolonged incubation time. In this study, the use of a flocked swab with BHI liquid medium resulted in a higher positivity rate and a significantly shorter time to positivity compared with the traditional cotton swab method. These findings suggest that the flocked swab culture method en- hances diagnostic efficiency in fungal keratitis.
This study included 15 patients with clinically suspected fungal keratitis (Table 1). The cohort comprised 10 males and 5 females, with a mean age of 49.9 years. Contact lens wear was the most common identified cause of infection, followed by ocular trauma. This finding is consistent with previous reports indicating that contact lens use and trauma are the leading causes of fungal keratitis12. Fungal keratitis is char- acterized by an insidious onset and a prolonged course, often resulting in delayed diagnosis8. In this study, the mean duration of illness before initial diagnosis exceeded 60 days, reflecting challenges in obtaining laboratory diagnoses and the high frequency of false-negative results. These features highlight the need for more sensitive and rapid culture methods.
Fungal growth was detected in 3 cases (20.0%) using the conventional method and in 13 cases (86.7%) using the flocked swab method (Fig. 3). Agreement between the two methods was poor (Cohen's κ = 0.01), indicating that the flocked swab method identified additional positive cases missed by the traditional method (Table 2). This difference in the culture results was statistically significant (McNemar test, p = 0.00195). Previous studies have reported fungal culture positivity rates ranging from approximately 40% to over 80-15. The relatively low culture positivity rate with the conventional method compared with previous studies may be attributed to the small sample size, prolonged disease duration before sampling, and the low-biomass nature of prolonged fungal keratitis. Prior empirical antimicrobial treat- ment before referral may have also contributed to reduced culture yield.
The high positivity rate observed with the flocked swab method is likely attributable not only to differences in fiber structure but also to its enhanced absorption and release efficiency, improved recovery of organisms even from low-biomass samples, and improved microbial survival during transport. The microstructure of the flocked swab confers distinct advantages for culture collection. Unlike conventional cotton swabs with irregularly arranged fibers, flocked swabs feature uniformly aligned fibers that facilitate efficient capture through capillary action21. Flocked swabs also demonstrate superior uptake and release efficiency. Warnke et al. reported that flocked swabs recovered greater numbers of bacteria and fungi, even under conditions where only a small amount of fluid was available16. In addition, Goldfarb et al. found that flocked swabs achieved a higher detection rate in low-density samples22. These findings are consistent with our observation that all 10 samples that were culture-negative using the conventional method were positive when collected with the flocked swab. Thus, the flocked swabs enable more effective collection and release of small quantities of microorganisms, thereby improving overall culture yield.
In addition, the use of flocked swabs in combination with BHI liquid medium may help preserve microbial viability during transport. Freddi et al. demonstrated higher Brucella survival when flocked swabs were stored in a protective medium23. The BHI liquid medium used in our study may have served a similar protective function. Its osmotically stable environment likely maintained fungal viability and minimized organism loss during transport. Moreover, BHI contains nutrients, such as proteins, peptones, and carbohydrates, that promote early fungal growth, even when the initial specimen quantity is small. Together, these factors likely account for the superior performance of the flocked swab combined with the BHI liquid medium culture method.
The median time to culture positivity was 96 hours with the flocked swab method and 116 hours with the conventional method (p = 0.016) (Fig. 4). The faster growth observed in the flocked swab group may reflect more efficient specimen recovery and rapid proliferation in liquid medium. Previous studies have likewise demonstrated that flocked swabs en- hance early colony formation19,21. These findings support the use of the flocked swab method to enable earlier diagnosis and more timely treatment decisions in clinical practice.
Among the three cases of mixed bacterial and fungal in- fections, two were missed by the conventional method but detected using the flocked swab method. Previous studies have shown that mixed bacterial-fungal infections are often difficult to culture because of interspecies competition, dif- fering growth requirements, and biofilm formation24. The higher detection of mixed bacterial and fungal infections with the flocked swab method suggests that this approach may improve the culture positivity rate even in polymicrobial infections.
This study has several limitations. First, it was conducted at a single center and included a relatively small sample size. Second, in this study, species-level identification was not performed, limiting pathogen-specific interpretation of the results. Future studies incorporating molecular or proteomic identification methods, such as internal transcribed spacer sequencing or matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS), may further enhance diagnostic accuracy. Third, since the flocked swab and BHI liquid medium were applied simultaneously in this study, the independent contribution of each component to the improved culture yield cannot be determined. Never- theless, these components may play complementary roles: flocked swabs enhance specimen uptake and release efficiency, while liquid enrichment media, such as BHI, may promote microbial survival and early growth, particularly when the initial organism burden is low. Future multicenter prospective studies with larger sample sizes are warranted to confirm these findings and refine culture-based diagnostic workflows for fungal keratitis.
In summary, the culture method using a flocked swab combined with the BHI liquid medium culture method demon- strated a higher positivity rate and a shorter time to positivity than the conventional cotton swab method. This approach may reduce the false-negative rate associated with conven- tional techniques and facilitate an earlier diagnosis of fungal keratitis.
References
1. 1. Mun Y, Kim MK, Oh JY. Ten-year analysis of micro- biological profile and antibiotic sensitivity for bacterial keratitis in Korea. PLoS ONE 2019;14:e0213103
Google Scholar
2. 2. Lin A, Rhee MK, Akpek EK, Amescua G, Farid M, Garcia-Ferrer FJ, et al. Bacterial keratitis preferred practice pattern®. Ophthalmology 2019;126:P1-P55
Google Scholar
3. 3. Prajna NV, Krishnan T, Rajaraman R, Patel S, Shah R, Srinivasan M, et al. Predictors of corneal perforation or need for therapeutic keratoplasty in severe fungal keratitis: A secondary analysis of the mycotic ulcer treatment trial II. JAMA Ophthalmol 2017;135:987-991
Google Scholar
4. 4. Prajna NV, Krishnan T, Mascarenhas J, Srinivasan M, Oldenburg CE, Toutain-Kidd CM, et al. Predictors of outcome in fungal keratitis. Eye (Lond) 2012;26:1226-1231
Google Scholar
5. 5. Prajna NV, Srinivasan M, Mascarenhas J, Lalitha P, Rajaraman R, McClintic S, et al. Visual impairment in fungal versus bacterial corneal ulcers four years after successful antimicrobial treatment. Am J Ophthalmol 2019;204:124-129
Google Scholar
6. 6. Colby K, Dana R. Foundations of corneal disease: Past, present and future. New York: Springer Nature; 2019
Google Scholar
7. 7. Kalkanci A. Microbiological diagnosis of fungal keratitis. In: Kalkanci A, editor. Mycotic keratitis. Florida, United States: CRC Press; 2019:45-55
Google Scholar
8. 8. Awad R, Ghaith AA, Awad K, Mamdouh Saad M, Elmassry AA. Fungal keratitis: Diagnosis, management, and recent advances. Clin Ophthalmol 2024;18:85-106
Google Scholar
9. 9. Menard M, Shah YS, Stroh IG, Zafar S, Sriparna M, Zhang N, et al. Microbial profile and clinical outcomes of fungal keratitis at a single-center tertiary care hospital. Clin Ophthalmol 2022;16:389-399
Google Scholar
10. 10. Ting DSJ, Gopal BP, Deshmukh R, Seitzman GD, Said DG, Dua HS. Diagnostic armamentarium of infectious keratitis: A comprehensive review. Ocul Surf 2022;23:27-39
Google Scholar
11. 11. Hare J. Sabouraud agar for fungal growth protocols. American Society for Microbiology 2008:1-5
Google Scholar
12. 12. Atta S, Perera C, Kowalski RP, Jhanji V. Fungal keratitis: Clinical features, risk factors, treatment, and outcomes. J Fungi 2022;8:962
Google Scholar
13. 13. Yarimada S, Barut Selver O, Palamar M, Egrilmez S, Aydemir SS, Hilmioğlu Polat S, et al. Comparison of culture-positive and -negative microbial keratitis. Turk J Ophthalmol 2022;52:1-5
Google Scholar
14. 14. Lin Y, Zhang J, Han X, Hu J. A retrospective study of the spectrum of fungal keratitis in southeastern China. Ann Palliat Med 2021;10:9480-9487
Google Scholar
15. 15. Mohd-Tahir F, Norhayati A, Siti-Raihan I, Ibrahim M. A 5-year retrospective review of fungal keratitis at hospital universiti Sains Malaysia. Interdiscip Perspect Infect Dis 2012;2012:851563
Google Scholar
16. 16. Warnke P, Warning L, Podbielski A. Some are more equal - a comparative study on swab uptake and release of bacterial suspensions. PLoS ONE 2014;9:e102215
Google Scholar
17. 17. Tananuvat N, Salakthuantee K, Vanittanakom N, Pongpom M, Ausayakhun S. Prospective comparison between conventional microbial work-up vs PCR in the diagnosis of fungal keratitis. Eye (Lond) 2012;26:1337-1343
Google Scholar
18. 18. Kuo MT, Chang HC, Cheng CK, Chien CC, Fang PC, Chang TC. A highly sensitive method for molecular diagnosis of fungal keratitis: Dot hybridization assay. Ophthalmology 2012;119:2434-2442
Google Scholar
19. 19. Daley P, Castriciano S, Chernesky M, Smieja M. Com- parison of flocked and rayon swabs for collection of respiratory epithelial cells from uninfected volunteers and symptomatic patients. J Clin Microbiol 2006;44: 2265-2267
Google Scholar
20. 20. Kumarajith TM, Breadmore M, Powell SM. Performance evaluation of commercially available swabs for environ- mental monitoring: Uptake and release efficiency. J Microbiol Methods 2024;216:106866
Google Scholar
21. 21. Van Horn KG, Audette CD, Tucker KA, Sebeck D. Com- parison of 3 swab transport systems for direct release and recovery of aerobic and anaerobic bacteria. Diagn Microbiol Infect Dis 2008;62:471-473
Google Scholar
22. 22. Goldfarb DM, Slinger R, Tam RK, Barrowman N, Chan F. Assessment of flocked swabs for use in identification of streptococcal pharyngitis. J Clin Microbiol 2009;47:3029-3030
Google Scholar
23. 23. Freddi L, Djokic V, Petot-Bottin F, Girault G, Perrot L, Ferreira Vicente A, et al. The use of flocked swabs with a protective medium increases the recovery of live Brucella spp. and DNA detection. Microbiol Spectr 2021; 9:e0072821
Google Scholar
24. 24. Little W, Black C, Smith AC. Clinical implications of poly- microbial synergism effects on antimicrobial susceptibility. Pathogens 2021;10:144
Google Scholar
Congratulatory MessageClick here!